This product has aleady completed the European In Vitro Diagnostic Medical Devices Dirctive (IVDD) compliance statement and registration in the European Union, and has been Qualified for EU market access.
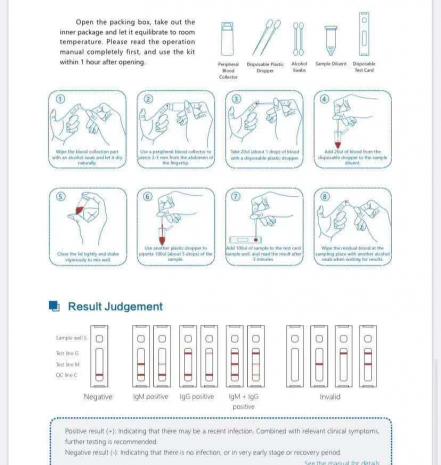
COVID-19 (SARS-COV-2) IGM/IGG Antibody Fast Detection Kit ( Colloidal Gold) adopts colloidal gold immunochromatography assay (GICA) . The test card contains 1.) colloidal gold labeled antigen and quality control antibody complex, 2.) Nitrocellulose membranes immobilized with two test lines (M line and G line) and one quality control line (C line).
Contact: Herry
NanJing KOZO Optical and Electronical Instrument Co.,Ltd
Sales Dep: 19E, DongYu Building, 18 ZhengHong Street, NanJing
Factory Add: A20 MuFu, 199 East Road MuFu, GuLou District, NanJing
Code: 210005 WeChat: 13357725973
Tel: 0086-25-84716665, 84715655, 84715659
Fax: 0086-25-84715556
Mail: sales@kozo-optics.com